The appropriate management of pain in the field has been discussed since the inception of the Wilderness Medical Society (WMS). The WMS first published its practice guidelines in book form in 1987. In 2014 the WMS published clinical practice guidelines for the management of pain in austere environments in the peer reviewed medical literature. Because intravenous (IV) access in the field can be problematic, discussions focused on the transmucosal delivery of opiates, particularly transmucosal immediate-release fentanyl (TIRF) medications. Fentanyl is a synthetic µ receptor-stimulating opioid. Fentanyl has become a popular analgesic in emergency medicine because it has minimal cardiovascular effects, does not increase plasma histamine, and is relatively short in onset of action and duration of effect. Transmucosal fentanyl preparations that have been developed over the last 25 years include oral transmucosal lozenges, buccal tablets, nasal spray, buccal soluble film, sublingual spray and sublingual tablets.
The military popularized the use for TIRF medications for on-the-battlefield analgesia and an application was proposed for wilderness and austere medicine in general. There was discussion about taping a fentanyl oral transmucosal lozenge (“lollipop”) to the finger of an individual in pain, where oversedation would cause their finger to drop away from their mouth resulting in automatic titration of dosing. Most realize now that the obtundation required for this automatic titration may result in significant respiratory suppression and hypoxia as most of these patients would be opioid naïve.
In 2011, the Food and Drug Administration (FDA) required a Risk Evaluation and Mitigation Strategy (REMS) to ensure that only individuals who were opioid tolerant were receiving TIRF medications. This required that healthcare providers who prescribed TIRF medications for outpatient use and pharmacies that dispensed them to be certified. Patients were also required to sign an agreement to participate in the REMS program. In December of 2022, the FDA approved a modified TIRF REMS because they became aware that opioid-naïve patients were still receiving TIRF medications. Healthcare providers who prescribe outpatient TIRF medications and pharmacies that dispense must still be certified in the TIRF REMS program, but providers must now document opioid tolerance with every outpatient prescription, and pharmacies must assess opioid tolerance with every dispensing. All outpatients receiving TIRF medications must now be enrolled in a registry. Despite the demonstrated safety and efficacy of TIRF medications in the field, this effectively precludes the carrying of and administration of TIRF medications in wilderness rescue.
When the patient is obese, a small child, in shock, the environmental conditions are adverse, or the individual starting the IV does not perform the procedure routinely (most healthcare providers), intravenous access can be challenging to obtain. For this reason, intraosseous access (IO) has been proposed as an alternative. Although IV or IO access is ideal for fluid resuscitation, few wilderness EMS (WEMS) teams carry multiple liters of crystalloid in the field. When fluid resuscitation is not possible or essential, wilderness rescuers should not overlook the oral route for analgesia. There are situations where patients in austere environments should have nothing by mouth: significant head injury, spinal cord injury, altered mental status, inability to protect the airway, or excessive nausea and vomiting. However, most patients, including those with sources of pain that will eventually need surgery, and especially those that will have long evacuations, will need oral hydration and calories to sustain themselves. Reduced oral intake of fluid predisposes to dehydration and calories fuel the thermogenesis of shivering. A comprehensive review by the Agency for Healthcare Research and Quality has concluded that acetaminophen (paracetamol) and non-steroidal anti-inflammatory drugs (NSAIDs) are not inferior to opioids for appropriate pain relief in the pre-hospital setting.
Now that TIRF medications are no longer available for wilderness rescue, consider: (1) allowing patients to take oral fluids and calories to reduce the risk of dehydration and hypothermia; (2) maximizing PRICE therapy (protection, rest, ice, compression, and elevation); (3) having the patient take non-opioid medications orally to improve comfort and facilitate the evacuation; (4) administering oral opioid medications after the patient has failed non-opioid medications. These recommendations mirror those from the American College of Emergency Physicians and Wilderness Medical Society. Included below are an algorithm showing an evidence-based basic life support (BLS) approach to managing pain in an austere environment and the pain management pyramid from the WMS pain management guidelines.
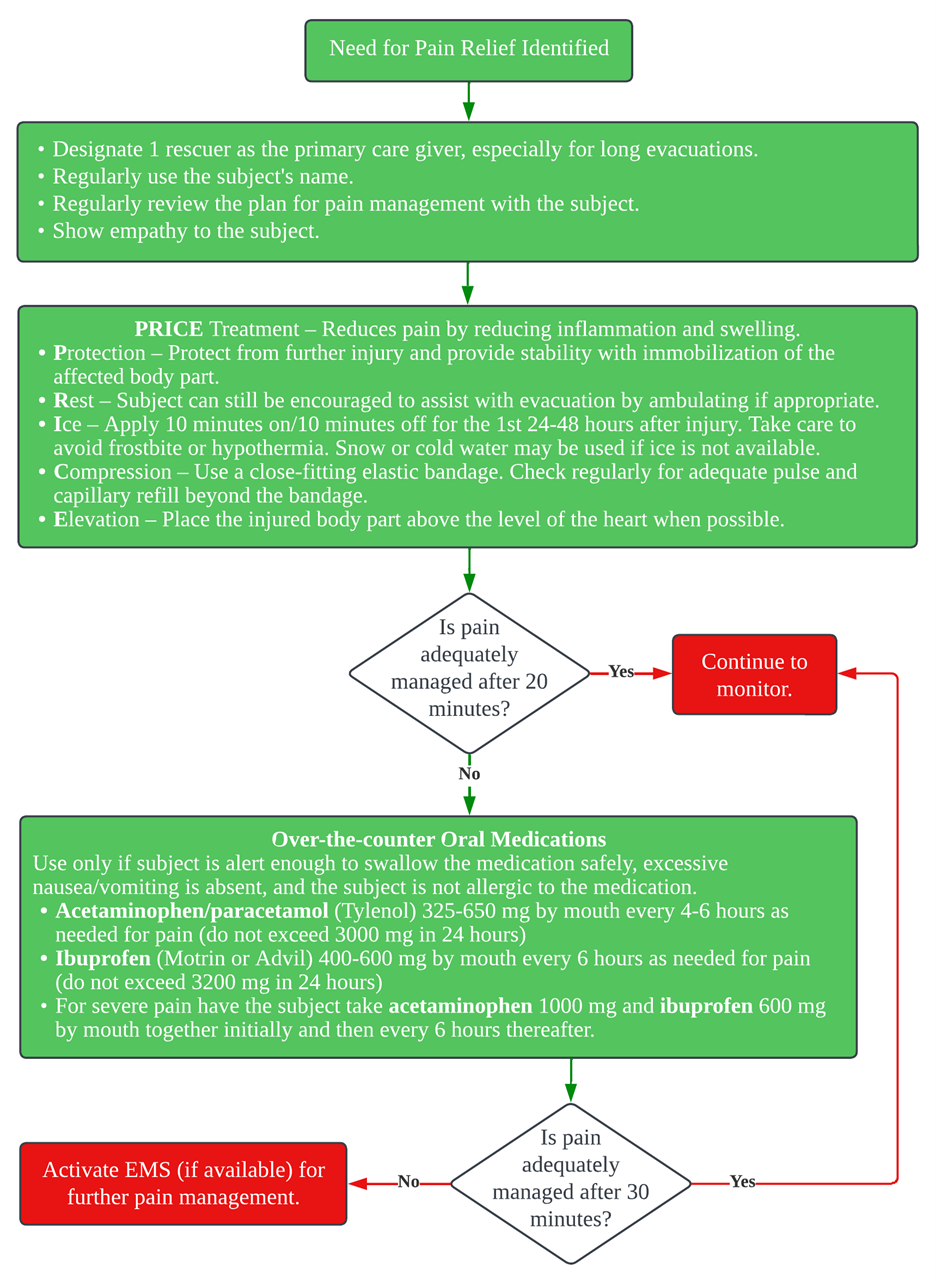
Pain relief flow chart (NIH).
Wilderness Medical Society practice guidelines for the treatment of acute pain in remote environments:
